Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 O
O thin films on Si (100) by ion beam sputter deposition
thin films on Si (100) by ion beam sputter depositionMao, W.*; Fujita, Masaya*; Chikada, Takumi*; Yamaguchi, Kenji; Suzuki, Akihiro*; Terai, Takayuki*; Matsuzaki, Hiroyuki*
Surface & Coatings Technology, 283, p.241 - 246, 2015/12
Times Cited Count:3 Percentile:13.89(Materials Science, Coatings & Films)Single-phase nanocrystalline thin films of Er O
O (440) has been first prepared using Si (100) substrates by ion beam sputter deposition at 973 K at a pressure of
(440) has been first prepared using Si (100) substrates by ion beam sputter deposition at 973 K at a pressure of  10
10 Pa and
Pa and 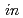 -
-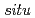 annealing at 1023 K at a pressure of
annealing at 1023 K at a pressure of  10
10 Pa. Er silicides formed during the deposition are eliminated via the annealing, which results in the single phase and the smooth surface of the Er
Pa. Er silicides formed during the deposition are eliminated via the annealing, which results in the single phase and the smooth surface of the Er O
O thin films. The epitaxial relationship between Si (100) and Er
thin films. The epitaxial relationship between Si (100) and Er O
O (110) is clarified by X-ray diffraction and reflection high energy electron diffraction.
(110) is clarified by X-ray diffraction and reflection high energy electron diffraction.
 O
O (001) surface; A DFT study
(001) surface; A DFT studyMao, W.*; Chikada, Takumi*; Shimura, Kenichiro*; Suzuki, Akihiro*; Yamaguchi, Kenji; Terai, Takayuki*
Journal of Nuclear Materials, 443(1-3), p.555 - 561, 2013/11
Times Cited Count:3 Percentile:25.73(Materials Science, Multidisciplinary)In this work,  calculations based on density functional theory (DFT) and generalized gradient approximation were performed to investigate the structural and electronic properties of the cubic Er
calculations based on density functional theory (DFT) and generalized gradient approximation were performed to investigate the structural and electronic properties of the cubic Er O
O (001) surface and H adsorption processes on this surface. Several stable adsorption sites were identified, and at the most energetically favorable adsorption sites it was found that H bonds with O atoms at the cubic Er
(001) surface and H adsorption processes on this surface. Several stable adsorption sites were identified, and at the most energetically favorable adsorption sites it was found that H bonds with O atoms at the cubic Er O
O (001) surface with an adsorption energy of 295.68 kJ mol
(001) surface with an adsorption energy of 295.68 kJ mol at coverage 1/8 ML, which was inclined to decrease with the increase of H coverage (
at coverage 1/8 ML, which was inclined to decrease with the increase of H coverage ( 1/4 ML). In addition, the calculations revealed that the dissociative H atom configurations have adsorption energies that are at least 152.64 kJ mol
1/4 ML). In addition, the calculations revealed that the dissociative H atom configurations have adsorption energies that are at least 152.64 kJ mol greater than the H
greater than the H molecule configurations on the surface. These results are discussed in regard of the hydrogen isotope permeation behavior in the tritium permeation barrier in a fusion reactor.
molecule configurations on the surface. These results are discussed in regard of the hydrogen isotope permeation behavior in the tritium permeation barrier in a fusion reactor.
 ErRuO
ErRuO
Izumiyama, Yuki*; Doi, Yoshihiro*; Wakeshima, Makoto*; Hinatsu, Yukio*; Nakamura, Akio; Ishii, Yoshinobu
Journal of Solid State Chemistry, 169(1), p.125 - 130, 2002/11
Times Cited Count:41 Percentile:81.15(Chemistry, Inorganic & Nuclear)no abstracts in English
Nakano, Yoshihiro; Ando, Masaki; Okajima, Shigeaki; Takano, Hideki; Akie, Hiroshi
Progress in Nuclear Energy, 38(3-4), p.343 - 346, 2001/02
Times Cited Count:1 Percentile:12.01(Nuclear Science & Technology)no abstracts in English
 /Si studied by slow positron beam
/Si studied by slow positron beamKawasuso, Atsuo; Arai, H.*; Hirata, K.*; Sekiguchi, T.*; Kobayashi, Yoshinori*; Okada, Sohei
Radiation Physics and Chemistry, 58(5-6), p.615 - 619, 2000/06
Times Cited Count:3 Percentile:26.42(Chemistry, Physical)no abstracts in English
 -doped silica-core optical fiber
-doped silica-core optical fiber*; *; *; *; Kusama, Yasuo; Seguchi, Tadao
Japanese Journal of Applied Physics, 33(7A), p.3937 - 3941, 1994/07
Times Cited Count:2 Percentile:17.88(Physics, Applied)no abstracts in English
 -rays induced loss in erbium-doped fiber
-rays induced loss in erbium-doped fiber*; *; *; *; Kusama, Yasuo; Seguchi, Tadao
DEI-93-165, 0, p.11 - 19, 1993/12
no abstracts in English
; ; ; ; *
Bunseki Kagaku, 15(6), p.589 - 594, 1966/00
no abstracts in English